Molecular screening techniques for multidrug-resistant organisms (MDRO) can reduce mortality rates and the economic burden of antimicrobial resistance (AMR)
Multidrug-resistant organisms (MDROs) are microorganisms (mainly bacteria) that become resistant due to antibiotic misuse as well as through natural mutations.1 Evidence exists that antimicrobial resistance (AMR) is one of the greatest threats to global health, food security and development in today’s society.1 AMR not only prevents the treatment of infectious diseases, such as pneumonia, but according to the World Health Organization (WHO) it also jeopardises the outcomes of modern medicine, as surgeries become much more dangerous without effective antibiotics.1
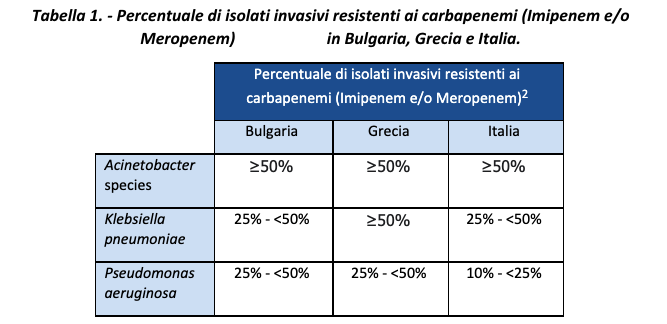
A European surveillance report published in 2020, based on data for 2015-2019, showed that European countries had up to 46.7% of invasive isolates of methicillin-resistant Staphylococcus aureus (MRSA).2 Particularly high rates of resistance to carbapenems were also found, especially in some countries including Greece, Italy and Bulgaria (Table 1).2
Percentage of invasive isolates resistant to carbapenems (Imipenem and/or Meropenem)
Between 2015 and 2019, in various European countries such as Ireland, Germany, Romania, Hungary, Greece, Latvia, Croatia, Lithuania, Poland, and Slovakia, a significant increase was also observed in the Vancomycin-resistant enterococci (VRE) with 25-50% of invasive isolates with resistance.2 Although Norway has one of the lowest rates of AMR in bloodstream infections, resistance to broad-spectrum antibiotics used to combat MDROs has increased in the last10 years.3 Looking specifically at the Italian context, the results of the national AR-ISS surveillance report published at the end of 2021 on the data collected in 2020 at the height of the COVID-19 pandemic still showed high rates of resistance to major antibiotic classes for the 8 pathogens under surveillance, with particular concern for MRSA (it remained stable at around 34%) and Enterococcus faecium (increasing the percentage of vancomycin-resistant isolates to 23.6%).4
The increase in MDRO prevalence across Europe thus requires effective intervention to reduce the social burden. Several risk factors influence the prevalence of MDROs (some of which emerged recently), which have a big impact on European health systems and communities. This negative impact is likely also due to the shortcomings in current screening techniques, resulting in a lack of detection and control of antimicrobial resistance.
Risk factors for MDROs in Europe
The best known risk factor for antimicrobial resistance is “antibiotic misuse”; globally, there is a large amount of antibiotic self-medication due to over-the-counter access. According to the European Centre for Disease Prevention (ECDC) and the WHO, Greece showed a higher defined daily dose (DDD) of antibiotic use in Europe and a 2.4% increase in consumption between 2014 and 2018, while many European countries saw a decrease.4 This, coupled with the high levels of antibiotic resistance in Greece, suggests a causal link.2
On the other hand, Italy (a country with high levels of antibiotic resistance) showed a relatively low DDD, suggesting the involvement of additional factors. New strains of MRSA were detected in Europe in 2021; in particular, clone t304/ST6 was identified in five northern European countries as originating from the Middle East.6 Moreover, a study of asylum seekers and refugees admitted to Helsinki University Hospital between 2010 and 2017 found that 45% were colonised by MDRO and 12.5% had two or more detectable MDRO strains.7 The percentage of MDRO carriers was highest amongst patients from Iraq (57.2%), Syria (55.8%), Afghanistan (34.8%) and Somalia (25.8%).7 Mass migration from low- and middle-income countries in the Middle East and Africa is prevalent in today's society, with Europe receiving 9.2 million asylum applications between 2010 and 2019.8 According to the European Commission, Italy and Greece are among the European countries with the highest number of first-time asylum applications.9
A recent study also found an association between MRSA and climate change, suggesting that a 1°C increase in average temperature could result in a 1.02-fold increase.10 There appears to be an association between average warm season temperature and MRSA detection, with a correlation coefficient of 0.826.10 Furthermore, it is estimated that the prevalence of carbapenemase-resistant carbapenemase-resistant Pseudomonas aeruginosa (CRPA) will double in the United Kingdom and the Netherlands by 2039 and increase by 70% in Denmark due to climate change alone.11 Another study in the United States found that a 10°C increase in temperature between regions caused an increase in antibiotic resistance of 4.2%, 2.2% and 2.7% for Escherichia coli, Klebsiella pneumoniae and Staphylococcus aureus; this increase was also associated with higher population density, suggesting that the growth of antimicrobial resistance should not be underestimated in currently growing populations.11
The impact of AMR on European countries and planned enforcement actions in Italy.
Lack of sufficient intervention to combat MDROs could lead to a 72% increase in second-line antibiotic resistance by 2030 in the EU/EEA, and last-line antibiotic resistance is likely to double.12 This has serious implications for European countries as infections lead to longer hospital stays, higher rates of antimicrobial resistance, higher medical costs, and increased mortality.13
Where first-line antibiotics can no longer be used, more expensive drugs are often required, placing a greater economic burden on families and society as a whole.1 In 2019, AMR and its ineffective treatment cost EU/EEA healthcare systems €1.1 billion.12 Alarmingly, in 2019, deaths in Europe due to AMR increased to 33,000.12 A rapid risk assessment for carbapenemase-resistant Enterobacteriaceae (CRE) in 2018 suggested that timely and appropriate laboratory investigations and reporting are essential to avoid delays in appropriate treatment and minimise the associated risk of death.15
In Italy, the 2022-2025 New National Antibiotic Resistance Plan, drafted by the Ministry of Health and recently published16, is structured around three main areas of action which involve a multidisciplinary and One Health approach (main element of innovation compared to previous plans):
- integrated surveillance and monitoring of AMR, antibiotic use, healthcare-associated infections (HAIs) and environmental monitoring;
- preventing HAIs in hospital and community settings and infectious diseases and zoonoses;
- appropriately using antibiotics in both human and veterinary settings and properly handling and disposing of antibiotics and contaminated materials.
Amongst the objectives in terms of AMR surveillance/monitoring are, for example, strengthening CRE surveillance, promoting the use of molecular methods. The importance of surveilling MDROs nationally had long been under consideration and had been reacknowledged as a necessity even by the AMCLI working group GLISTer in an article in 2017, with a focus on assisted living residences and assimilated local facilities, whilst also identifying three different levels of possible surveillance.
Molecular techniques as a way to supplement to classical cultural techniques
Traditionally, screening for antimicrobial resistance uses plate culture techniques, where the process of obtaining the result (TAT) from start to finish can take up to five days.18 Waiting this long for identification can lead to delays in implementing appropriate infection control measures, potentially leading to further transmission and mutationi. Delayed identification can also lead to delays in initiating the right treatment, which can also contribute to bacterial resistance and could be detrimental to the patient’s health or even prove fatal.
One study observed that when specifically comparing the detection of methicillin-sensitive S. aureus (MSSA) and MRSA using culture techniques versus a PCR test, detection is consistently underestimated. For culture methods, direct seeding detected 22.4% of MSSA and 1.2% of MRSA in the samples compared with 35.6% of MSSA and 2.3% of MRSA using a PCR assay.19 In the same study, they modelled these results on national surveillance data and estimated that about 5,000 to 8,000 surgical site infections of S. aureus could be prevented using molecular screening methods. In the UK alone, this could save between £17 million and £130 million per year in treatment costs.19
There are several issues (and their implications) in the research and development of an effective screening test for MDRO. Molecular diagnostics for MDRO can be useful as a second step to confirm phenotypic culture tests and in surveillance, for example, to confirm the mechanisms responsible for certain resistance20. However, culture methods are believed to heavily underestimate the number of MDROs within a sample, and such false negatives must be minimised to reduce the impact on healthcare systems. This suggests a use of molecular tests as a first step, but although they provide important and clinically relevant information, they themselves have limitations: in fact, molecular tests are more sensitive than culture tests, but they detect only known resistance genes or mutations, and phenotypic resistance testing (culture test) is always necessary in surveillance in the event of a positive result to ensure the correct classification of bacterial isolates20. In any case, it emerged that molecular and phenotypic diagnostics of MDROs complement each other to improve understanding of both the extent of resistance in a given context and the underlying mechanisms responsible for resistance20.
BD’s commitment and solutions
BD stands daily alongside healthcare practitioners around the world in the fight against AMR. BD is committed to helping slow the spread of antibiotic resistance by improving awareness, surveillance, infection prevention and patient management.
We offer a wide range of solutions that can be used to prevent the spread of infections in healthcare facilities, diagnostic systems for monitoring and diagnosing healthcare-associated infections (HAIs), including drug-resistant strains, and state-of-the-art surveillance and reporting capabilities to monitor and predict MDRO outbreaks.
More specifically:
- the range of validated molecular tests for the fully automated BD MAX™ system for monitoring HAIs and major MDROs (including MDR-TB) ensures early and accurate detection which, when combined with the right antimicrobial treatment, can prevent transmission and improve patient management. Learn more about the molecular tests available for CPO, MRSA, VRE and C. diff. [Link to the HAI Molecular Biology brochure/BD Academy information material]
- solutions for cultural and phenotypic analysis (manual and electronic), including the BD Phoenix™ M50 and related panels, associated with the BD Bruker MALDI BioTyper® and the BD Epicenter™ middleware which ensures accurate ID/AST results for correct microorganism classification and antibiotic prescription. Find out more HERE.
BD is an all-round partner in the MDRO surveillance workflow. Learn more about BD’s commitment to combating AMR and HAIs HERE.
BD-77376
Bibliography
- World Health Organization. Antibiotic Resistance. 2020. Available at: https://www.who.int/news-room/fact-sheets/detail/antibiotic-resistance. Accessed: July 2021.
- European Centre for Disease Prevention and Control. Surveillance report. Antimicrobial resistance in the EU/EEA (EARS-Net). 2019. Available at: https://www.ecdc.europa.eu/en/publications-data/surveillance-antimicrobial-resistance-europe-2019. Accessed: July 2021.
- European Centre for Disease Prevention and Control. ECDC country visit to Norway to discuss antimicrobial resistance issues. ECDC. 2019. Available at: https://www.ecdc.europa.eu/sites/default/files/documents/antimicrobial-resistance-country-visit-norway.pdf. Accessed: July 2021.
- Epicenter – AR-ISS Surveillance System Report. Available at: https://www.epicentro.iss.it/antibiotico-resistenza/ar-iss/RIS-1_2021.pdf – Accessed: Oct 2022.
- Robertson J, Vlahović-Palčevski V, Iwamoto K, Högberg LD, Godman B, Monnet DL, Garner S, Weist K, Strauss R, Vandael E, and Ivanov IN. Variations in the Consumption of Antimicrobial Medicines in the European Region, 2014–2018: Findings and Implications from ESAC-Net and WHO Europe. Front Pharmacol. 2021;12:639207.
- Bartels MD, Worning P, Andersen LP, Bes M, Enger H, Ås CG, Hansen TA, Holzknecht BJ, Larssen KW, Laurent F, and Mäkitalo B. Repeated introduction and spread of the MRSA clone t304/ST6 in northern Europe. Clin Microbiol Infect. 2021;27(2):284.e1-284.e4.
- Aro T and Kantele A. High rates of meticillin-resistant Staphylococcus aureus among asylum seekers and refugees admitted to Helsinki University Hospital, 2010 to 2017. Euro Surveill. 2018;23(45):1700797.
- United Nations High Commissioner for Refugees. Global trends: Forced Displacement in 2019. 2019. Available at: https://www.unhcr.org/5ee200e37.pdf. Accessed: July 2021.
- European Commission. Overall figures of immigrants in European society. 2020. Available at: https://ec.europa.eu/info/strategy/priorities-2019-2024/promoting-our-european-way-life/statistics-migration-europe_en. Accessed: July 2021.
- Kaba HE, Kuhlmann E, and Scheithauer S. Thinking outside the box: association of antimicrobial resistance with climate warming in Europe–a 30 country observational study. Int J Hyg Environ Health. 2020;223(1):151-158.
- MacFadden DR, McGough SF, Fisman D, Santillana M, and Brownstein JS. Antibiotic resistance increases with local temperature. Nat Clim Change. 2018;8(6):510-514.
- European Centre for Disease Prevention and Control. and Organisation for Economic Cooperation and Development. Antimicrobial Resistance: Tackling the Burden in the European Union. 2019; pp.3. Available at: https://www.oecd.org/health/health-systems/AMR-Tackling-the-Burden-in-the-EU-OECD-ECDC-Briefing-Note-2019.pdf. Accessed: July 2021.
- Váradi L, Luo JL, Hibbs DE, Perry JD, Anderson RJ, Orenga S, and Groundwater PW. Methods for the detection and identification of pathogenic bacteria: past, present, and future. Chem Soc Rev. 2017;46(16):4818-4832.
- European Centre for Disease Prevention and Control and the European Medicines Agency. The bacterial challenge: time to react. A call to narrow the gap between multidrug-resistant bacteria in the EU and the development of new antibacterial agents. Available at: https://www.ecdc.europa.eu/sites/portal/files/media/en/publications/Publications/0909_TER_The_Bacterial_Challenge_Time_to_React.pdf. Accessed: July 2021.
- European Centre for Disease Prevention and Control. Rapid Risk Assessment: Carbapenem-resistant Enterobacteriaceae – first update. 2018. Available at: https://www.ecdc.europa.eu/sites/default/files/documents/RRA-Enterobacteriaceae-Carbapenems-European-Union-countries.pdf. Accessed: July 2021.
- Aboutpharma drafting. Antibioticoresistenza: il nuovo Piano nazionale 2022-25 è all’esame delle Regioni. 21 September 2022. https://www.aboutpharma.com/animal-health/antibioticoresistenza-il-nuovo-piano-nazionale-2022-25-e-allesame-delle-regioni/
- Aschbacher, R., Pagani, L., Migliavacca, R. et al. Recommendations for the surveillance of multidrug-resistant bacteria in Italian long-term care facilities by the GLISTer working group of the Italian Association of Clinical Microbiologists (AMCLI). Antimicrob Resist Infect Control 9, 106 (2020). https://doi.org/10.1186/s13756-020-00771-0
- Scerbo MH, Kaplan HB, Dua A, Litwin DB, Ambrose CG, Moore LJ, Murray CCK, Wade CE, and Holcomb JB. Beyond blood culture and gram stain analysis: a review of molecular techniques for the early detection of bacteremia in surgical patients. Surg Infect (Larchmt). 2016;17(3):294-302.
- Tsang STJ, McHugh MP, Guerendiain D, Gwynne P, Boyd J, Walsh TS, Laurenson IF, Templeton KE, and Simpson AHRW. Underestimation of Staphylococcus aureus carriage associated with standard culturing techniques. Bone Joint Res. 2018;7(1):79-84.
- Global Antimicrobial Resistance Surveillance System (GLASS). Molecular methods for antimicrobial resistance (AMR) diagnostics to enhance the Global Antimicrobial Resistance Surveillance System. Geneva: World Health Organization; 2019 (WHO/WSI/AMR/2019.1). Licence: CC BY-NC-SA 3.0 IGO. https://www.who.int/publications/i/item/WHO-WSI-AMR-2019.1
